Cellular senescence, a dysfunctional state that aged cells enter, is considered one of the primary hallmarks of aging. But a lesser-known hallmark of aging is dysbiosis, the disruption of the microbiome that exists in your gut, mouth, and skin. While dysbiosis is primarily associated with the gut microbiome, a fascinating interplay exists between senescent skin cells and the bacteria that grow among them.[1]
What Happens to Your Skin Microbiome as You Age?
Multiple studies have documented that the skin microbiome undergoes significant shifts as it ages. In your youth, sebum-loving bacteria like Cutibacterium and Staphylococcus thrive, metabolizing oils, stabilizing pH, and outcompeting pathogens to maintain a low-inflammatory barrier.[2]
However, as the skin ages and accumulates damage from UV, pollution, and declining sebaceous output, these beneficial species wane.[2] Meanwhile, pro-inflammatory species flourish, fueled by a higher surface pH, increased TEWL (transepidermal water loss), chronic inflammation, and reduced antimicrobial peptides.[3] This can also be accelerated by events such as menopause, which exacerbate these skin changes that impact the microbiome.[4]
Notably, older adults often exhibit a higher diversity of skin microbes relative to younger adults, which some researchers suggest may result from reduced immune function, allowing more opportunistic species of bacteria and pathogens to spread.[2,5] This leads the skin microbiome to reflect the biological aging process. In fact, a recent study found that the skin microbiome can predict chronological age more accurately than the gut or oral microbiome.[6]
What is the Connection Between Skin Dysbiosis and Senescence?
Although skin dysbiosis and cellular senescence are each influenced by several aging factors, research supports a bidirectional and causational relationship between the two.
Does Skin Dysbiosis Drive Cellular Senescence?
The skin microbiome plays a critical role in educating your immune system to distinguish between harmless and harmful microbes. While a healthy microbiome modulates immune responses, its dysregulation can introduce chronic antigenic stress.[3,7] Essentially, the immune system becomes exhausted in attempting to fight off heightened levels of harmful microbes.
This persistent immune activation leads to the elevated production of free radicals by skin cells, which induces DNA damage and mitochondrial dysfunction, key drivers of cellular senescence. It's important to note that the relative newness of this science limits direct causal studies on this model.[3,8] However, some in vitro studies have shown that certain common bacteria, such as Pseudomonas aeruginosa, can secrete harmful compounds like pyocyanin that directly trigger senescence in skin cells.[9]
Additionally, dysbiosis can affect epidermal turnover and delayed barrier restoration. When these defects persist, they contribute to local inflammation that accelerates aging pathways.[3,8] Thus, microbial dysbiosis may act both as a trigger and amplifier of senescent cell accumulation in aged or compromised skin. Overall, the aging process is consistently accompanied by a shift toward a “dysbiotic” skin microbiome composition that potentially fosters more inflammation and reduced barrier function compared to youthful skin.[2,3,8]
Can Senescent Cells Worsen Skin Dysbiosis?
The interplay between senescence and dysbiosis also goes the other way. Senescent cells disrupt the homeostasis of your microbiome through both biochemical and structural changes.
SASP: The senescence-associated secretory phenotype, or SASP, is released by senescent cells as a signaling mechanism to your immune system to clear them away. Yet, if left to linger, the mix of inflammatory markers and collagen-degrading MMPs released can also induce nearby cells to enter a senescent state. At the same time, SASP increases skin pH and disrupts the skin's lipid matrix, potentially promoting bacteria that thrive in this pro-inflammatory environment.[3,8] Since an exhaustion of the immune system often accompanies this change, leaving senescent cells to linger for longer than they should, these harmful microbes often establish a more permanent residence on the skin, contributing to long-term, age-related skin issues.[3]
Structural Changes: By releasing enzymes that break down the skin's structural proteins, SASP has a significant impact on the skin's structural integrity. A weakened barrier means the skin's surface becomes drier, less acidic, and more prone to tiny micro-fissures in the epidermis, all of which can encourage opportunistic microbes to overgrow.[8] Thus, senescence-driven barrier damage can lead to colonization by less skin-friendly microbes, tilting the microbiome towards dysbiosis.
Through these mechanisms, senescent cells can trigger dysbiosis of the skin microbiome. Consequently, older skin with many senescent cells tends to be colonized by harmful rather than protective bacteria, especially when the barrier is impaired. An analysis of elderly skin microbiota revealed that skin changes associated with senescence cause beneficial bacteria, such as Cutibacterium, to decrease, while pathogens increase.[2]
Is Senescence a Root Cause of Skin Dysbiosis?
While initial dysbiosis can trigger senescence, there is substantial evidence that senescence may independently drive and maintain dysbiotic states. Specifically, the changes associated with senescence create a niche for pathogenic microbes, particularly those capable of thriving in high-pH, inflamed environments.
This fits well into the established model of the 12 hallmarks of aging, where there are three classes of pro-aging factors:
- Primary: These factors, like telomere shortening or damage to the genome, accumulate throughout life and contribute to other pro-aging effects.[1]
- Antagonist: As a response to the damage generated by primary hallmarks, the body triggers mechanisms like senescence that may initially have beneficial effects but play a detrimental role later in life. Senescence, for example, is thought to play a role in preventing the development of cancerous cells by sending an SOS signal to your immune system using SASP signals. Yet, when left to linger, senescence can be a key driver of aging.[1]
- Integrative: The last step in this chain, integrative hallmarks—like dysbiosis—are triggered by the accumulated damage of both primary and antagonist hallmarks.[1]
Based on this model, it is likely that senescent cells contribute significantly to the age-associated skin changes responsible for dysbiosis, meaning the microbiome shift is a downstream effect of cellular senescence.[1,2] However, the use of this model in this way does have its limitations. Because each hallmark of aging has at least some influence on the others, reducing the dimensionality of this process down to only 2 of the 12 hallmarks of aging may not account for the full scope of potential external influences.[1]
What is the Effect of This Microbial and Cellular Dysfunction on Skin?
The combined effects of cellular senescence and dysbiosis can exacerbate some of the most common skin concerns associated with aging. This includes wrinkles, dryness, hyperpigmentation, and a reduction in the skin's structural integrity. Yet this can have serious impacts on both the health of your skin and your skin's ability to repair itself:
Age-Related Skin Conditions
Certain dermatologic conditions of the elderly likely represent an exacerbation of the dysbiosis-senescence interaction.
Eczema: In older adults, atopic dermatitis often assumes a more stubborn form driven by both microbial imbalance and cellular aging. Triggered by a weakening of the skin barrier, which could be due to senescence, beneficial bacteria dwindle, and harmful bacteria colonize more readily. Together, they disrupt the skin barrier, keeping the skin stuck in a loop of inflammation, itching, and redness that’s hard to break without directly addressing both triggers.[10,11]
Photoaging: UV exposure induces cellular senescence in skin cells and also perturbs the local microbiome by killing specific beneficial bacteria and selecting for UV-tolerant microbes. Chronically sun-exposed skin harbors a distinct microbiome compared to sun-protected skin. This suggests that extrinsic aging factors (UV, pollution) can simultaneously induce senescence and dysbiosis, jointly contributing to wrinkles, hyperpigmentation, and other hallmarks of photoaging.[12]
Impaired Wound Healing
Delayed or non-healing wounds represent one of the most clinically significant outcomes of the dysbiosis–senescence loop. Chronic wounds often harbor biofilms composed of P. aeruginosa, S. aureus, and other opportunistic pathogens that exacerbate tissue inflammation and slow or prevent wound healing.[8]
Simultaneously, in poor healing wounds, senescent cells accumulate in the wound bed, releasing SASP that impair collagen synthesis, re-epithelialization, and angiogenesis. Some animal tests demonstrate that wound healing is markedly improved when senescent cells are removed or when microbial burden is reduced, supporting the concept that both must be addressed in tandem.[8,13]
Fight Back Against Skin Dysbiosis and Cellular Senescence With OS-01 FACE
Breaking free from the feedback loop of skin dysbiosis and cellular aging means attacking both problems head-on. Here's how OS-01 FACE does just that.
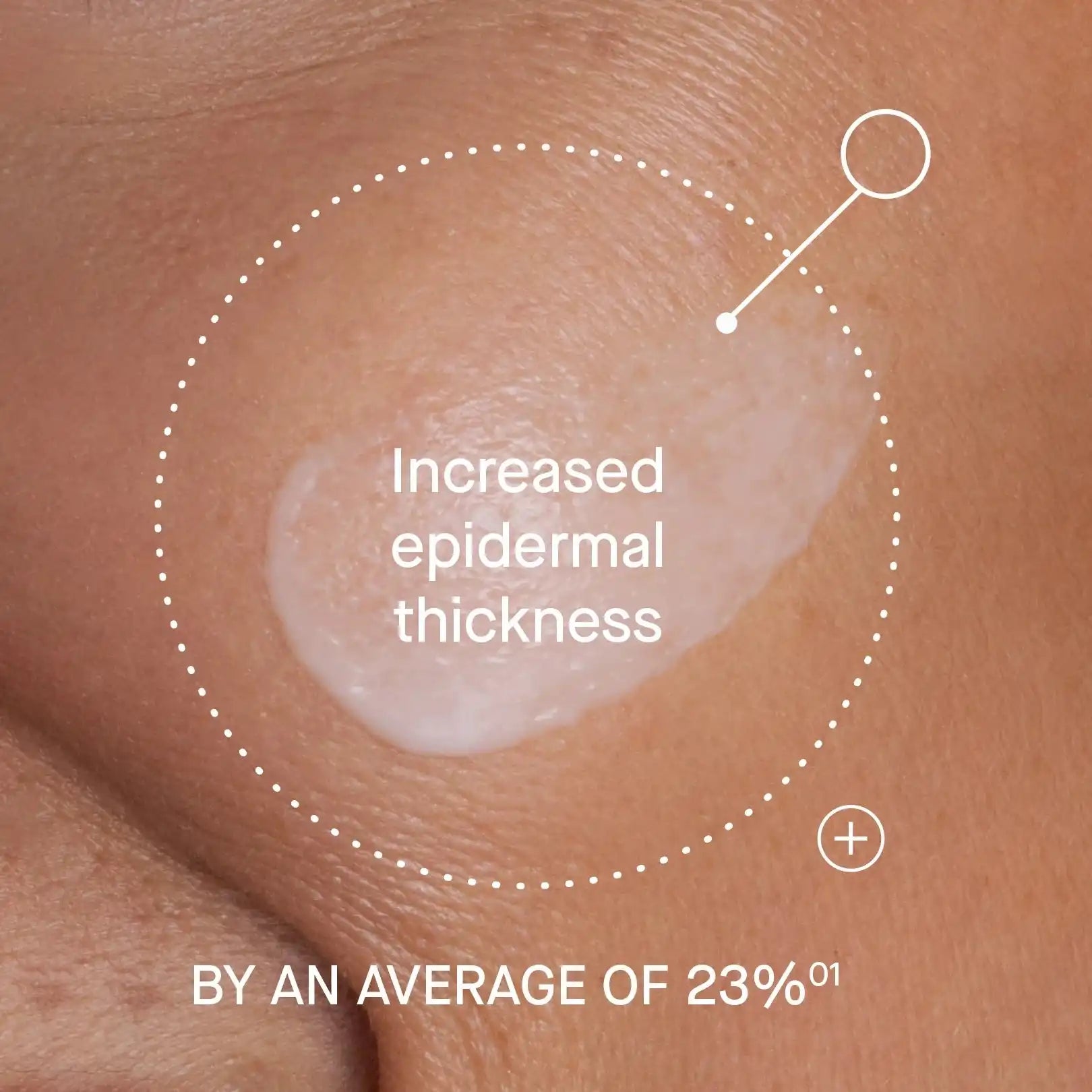
- Senescence: Our topical moisturizers, like OS-01 FACE, are formulated with the OS-01 peptide. In lab testing on human skin samples, the OS-01 peptide was shown to reduce cellular senescence, while curbing the expression of SASP. This helps prevent and reverse some of the signs of skin aging associated with senescence, like a weakened skin barrier. In fact, OS-01 FACE is clinically proven to improve skin barrier function by +15-19%.[14,15]
- Microbiome Balance: In a six-week clinical study, twice-daily application of OS-01 FACE significantly improved skin microbiome balance. This resulted in an increase in the relative abundance of beneficial bacteria, such as Cutibacterium, and a decrease in the diversity of other, potentially harmful, bacterial species – changes that were consistent with a younger skin microbiome profile.[16]
Key Takeaways
- The diversity and composition of the skin microbiome shift with age, reflecting age-related changes in skin pH, barrier function, and immune regulation.
- Senescent cells may be a root cause of dysbiosis by secreting SASP factors that trigger age-related skin changes, which in turn encourage microbial imbalance.
- Evidence suggests a bidirectional relationship between microbial imbalance and cellular senescence in the skin.
- Both microbial imbalance and senescence are linked to conditions like eczema and photoaging in older adults.
- In clinical and lab testing, OS-01 FACE improved skin microbiome balance and reduced markers of senescence.
References
- https://www.sciencedirect.com/science/article/pii/S0092867422013770
- https://microbiomejournal.biomedcentral.com/articles/10.1186/s40168-024-01891-0
- https://encyclopedia.pub/entry/24723
- https://pmc.ncbi.nlm.nih.gov/articles/PMC10991793/
- https://www.mdpi.com/2072-6643/13/12/4550
- https://pubmed.ncbi.nlm.nih.gov/32047061/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3535073/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC9963692/
- https://pubmed.ncbi.nlm.nih.gov/19286324/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC8198400/
- https://www.sciencedirect.com/science/article/abs/pii/S0923181124000677
- https://www.mdpi.com/1422-0067/22/8/3849
- https://pmc.ncbi.nlm.nih.gov/articles/PMC2984611/
- Zonari, A., et al. npj Aging, 2023
- Zonari, A., et al. Journal of Cosmetic Dermatology, 2024
- Based on data from clinical studies and/or lab studies conducted on human skin samples, 3D skin models, and skin or hair cells in the OneSkin lab. Explore more at oneskin.co/claims